AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Liquid matter9/15/2023  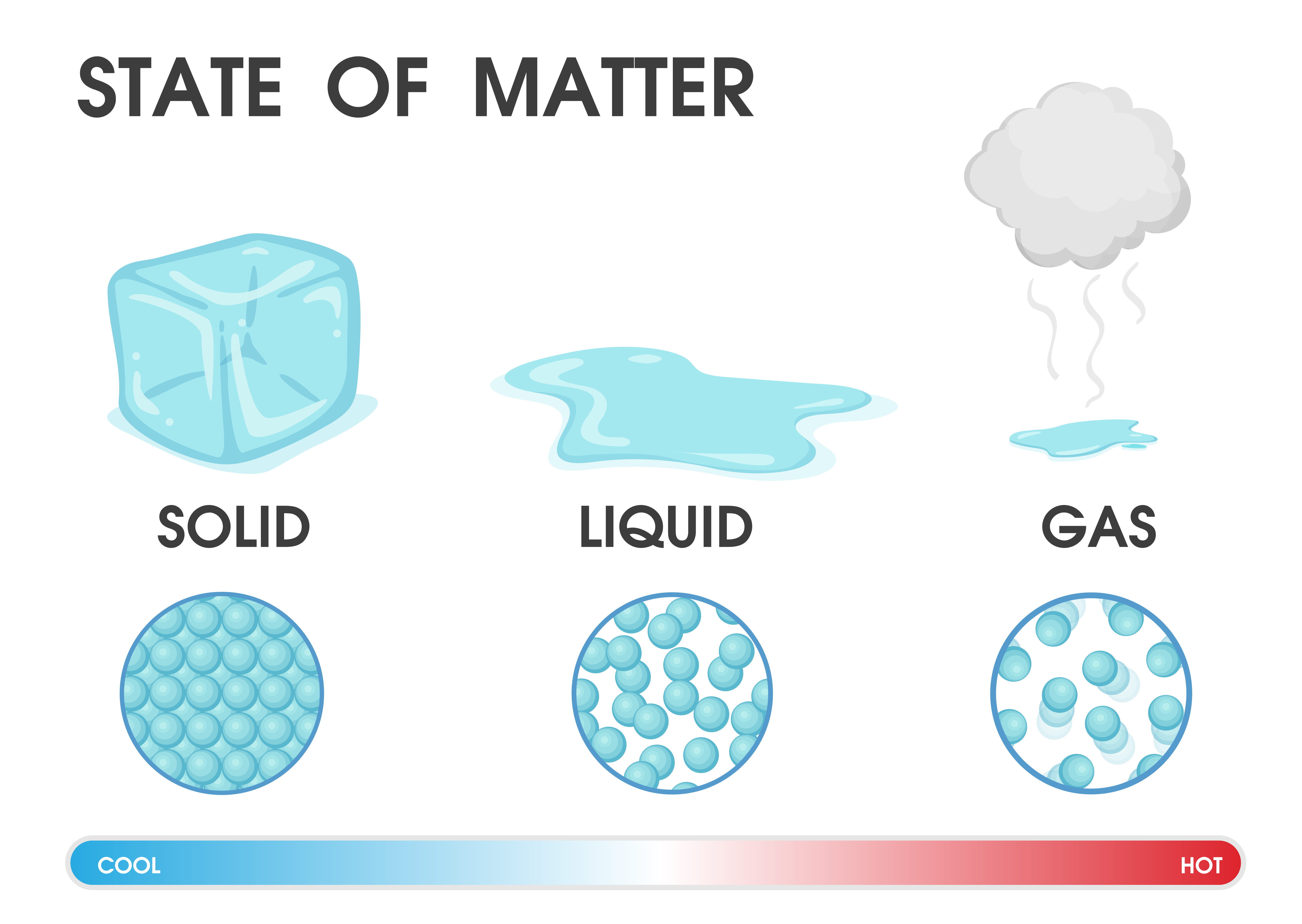
There are many different crystal structures, and the same substance can have more than one structure. In crystalline solids, particles are packed in a regularly ordered, repeating pattern. 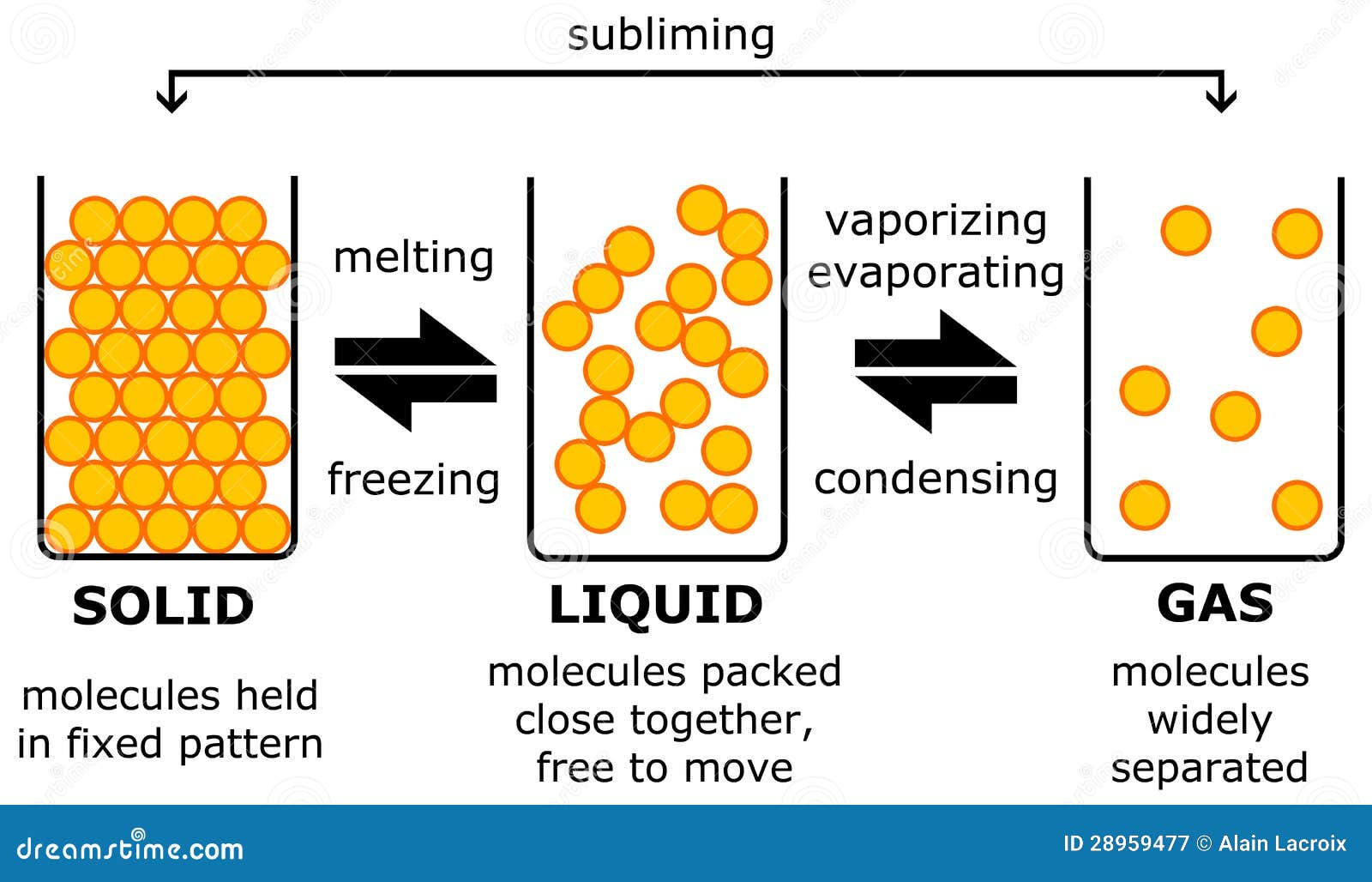
Solids can only change shape under force, as when broken or cut. As a result, a solid has a stable, definite shape and a definite volume. The forces between the particles are strong enough that the particles cannot move freely they can only vibrate. SolidsĪ solid’s particles are packed closely together. This diagram shows the nomenclature for the different phase transitions. Each of these three classical states of matter can transition directly into either of the other two classical states. Solid is the state in which matter maintains a fixed volume and shape, liquid is the state in which matter adapts to the shape of its container but varies only slightly in volume, and gas is the state in which matter expands to occupy the volume and shape of its container. Historically, the states of matter were distinguished based on qualitative differences in their bulk properties. Much of the atomic matter of the universe is hot plasma in the form of rarefied interstellar medium and dense stars. Further states, such as quark-gluon plasmas, are also believed to be possible. In extreme environments, other states may be present, such as plasma, Bose-Einstein condensates, and neutron stars. 
The three states of matter are the three distinct physical forms that matter can take in most environments: solid, liquid, and gas.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
